MUSCLE STRENGTH MAINTAINED
IN FIRDAPSE-TREATED PATIENTS1
The differences between those treated with FIRDAPSE and those treated with placebo were shown to be statistically significant based on Quantitative Myasthenia Gravis (QMG) scores in 2 phase 3 studies.1-3
Patients treated with FIRDAPSE maintained their muscle strength, while a statistically significant decline was demonstrated in patients in the placebo group.1-3
Coprimary Endpoint
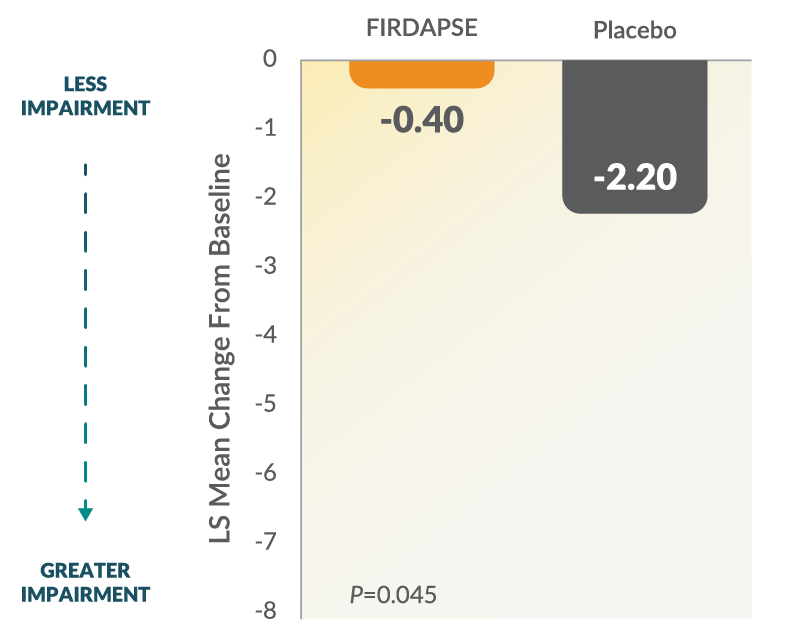
Study 1: Total QMG Changes From Baseline at Day 141,2
Study 2: Total QMG Changes From Baseline at Day 41,3
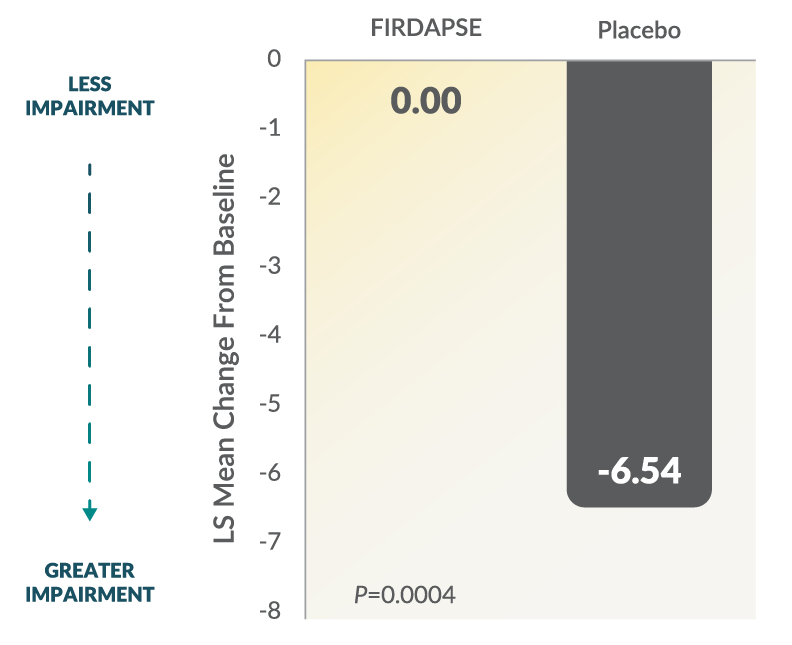
The QMG assessment is a 13-item, physician-rated categorical scale assessing muscle weakness. Each assessment is rated 0 to 3, where a score of 0 represents no weakness and a score of 3 represents severe weakness (total score ranges from 0 to 39). Higher scores represent greater impairment.1
PATIENT PERCEPTION OF PHYSICAL WELL-BEING MAINTAINED IN FIRDAPSE-TREATED PATIENTS1-3
The differences between those treated with FIRDAPSE and those treated with placebo were shown to be statistically significant based on Subject Global Impression (SGI) scores in 2 phase 3 studies.1-3
FIRDAPSE-treated subjects reported a statistically significant better sense of well-being compared to those in the placebo group.1-3
Coprimary Endpoint
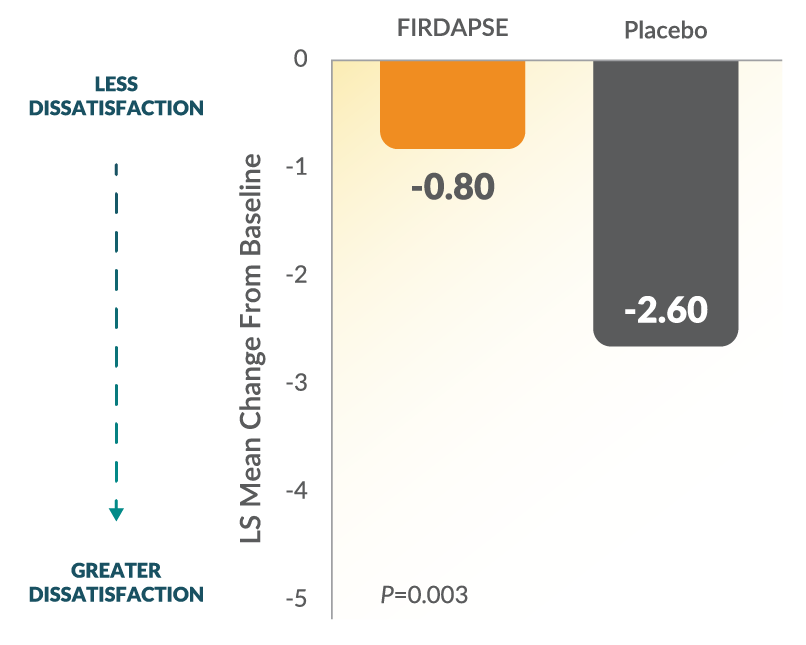
Study 1: SGI Changes From Baseline at Day 141,2
Study 2: SGI Changes From Baseline at Day 41,3
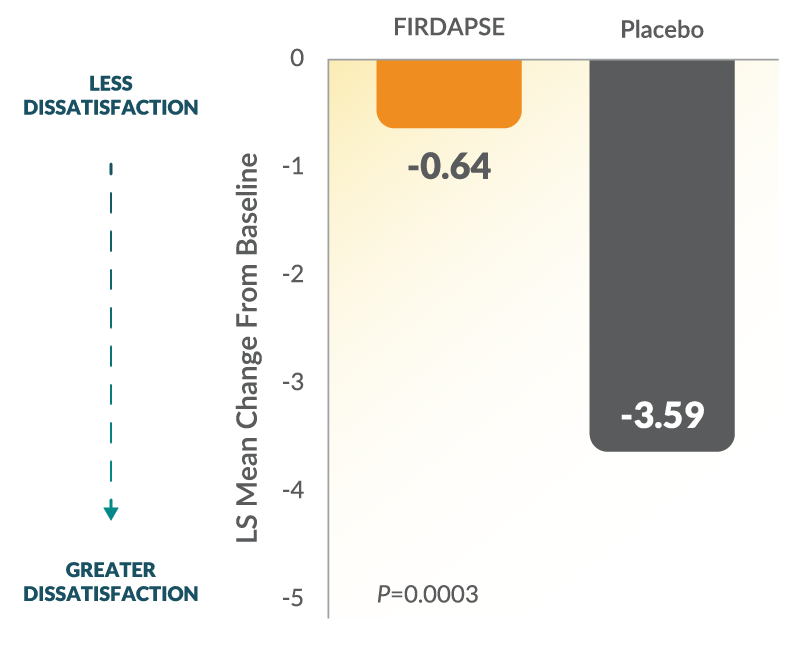
The SGI assessment is a 7-point scale on which patients rate their global impression of the study treatment effects on their physical well-being. Lower scores on the SGI represent lower perceived benefit with the study treatment. The 7-point SGI scale: 1=terrible, 2=mostly dissatisfied, 3=mixed, 4=partially satisfied, 5=mostly satisfied, 6=pleased, and 7=delighted.1-3
CLINICIAN IMPRESSION OF PATIENT WELL-BEING IMPROVED WITH FIRDAPSE1-3
The differences between those treated with FIRDAPSE and those treated with placebo were shown to be statistically significant based on Clinical Global Impression-Improvement (CGI-I) scores in 2 phase 3 studies.1-3
FIRDAPSE-treated subjects demonstrated a statistically significant improvement in symptoms, behavior, and functional ability compared with those in the placebo group.1,3
Secondary Endpoint
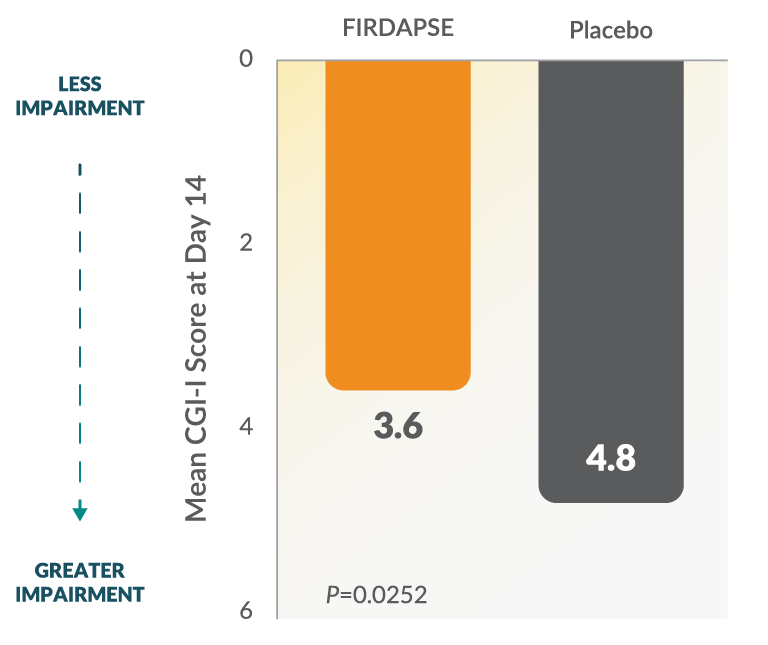
Study 1: CGI-I Scores at Day 141,2
Study 2: CGI-I Scores at Day 41,3
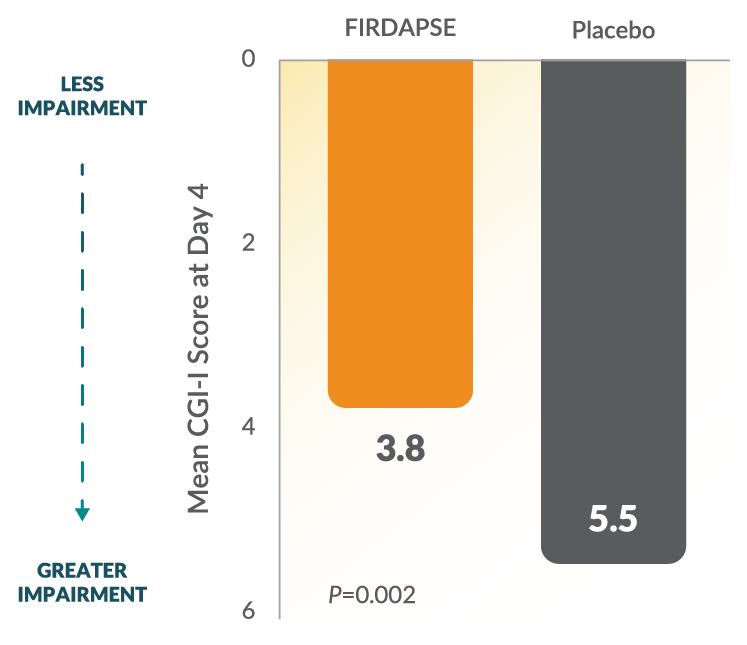
The CGI-I is a subjective, investigator-assessed overall impression of improvement or worsening in symptoms. The 7-point scale is scored from 1 (very much improved) to 7 (very much worse) based on symptoms, behavior, and functional ability. CGI-I scores were collected as a secondary endpoint during this study.26
FIRDAPSE PATIENTS MAINTAINED FUNCTIONAL MOBILITY3
The Triple-Timed Up-and-Go Test (3TUG) was an exploratory endpoint in a phase 3 study and showed the differences between placebo-treated subjects and those treated with FIRDAPSE.3,27,28
The proportion of patients with a ≥20% increase in the 3TUG average time was higher in the placebo group compared with the proportion of patients in the FIRDAPSE group.3
Exploratory Endpoint
Study 2: 3TUG Average Time3
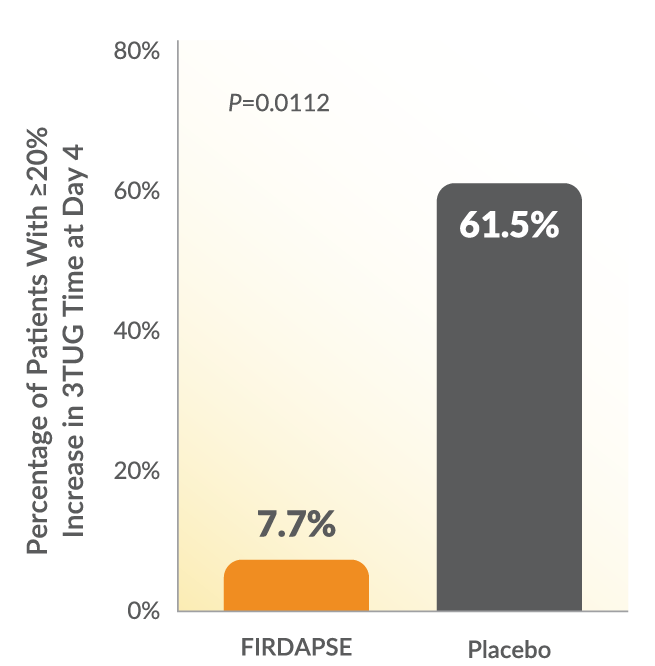
The 3TUG is a functional mobility test that requires a patient to stand up from a straight-backed armchair, walk 3 meters, turn around, walk back, and sit down in the chair. Individuals perform the test 3 consecutive times without pause, and the final measurement is the average time required to complete all 3 repetitions. Based on literature reports, a significant change in gait for a similar walk test is an increase in time of >20%, which was incorporated into the endpoint.3
TRIAL 1 STUDY DESIGN2
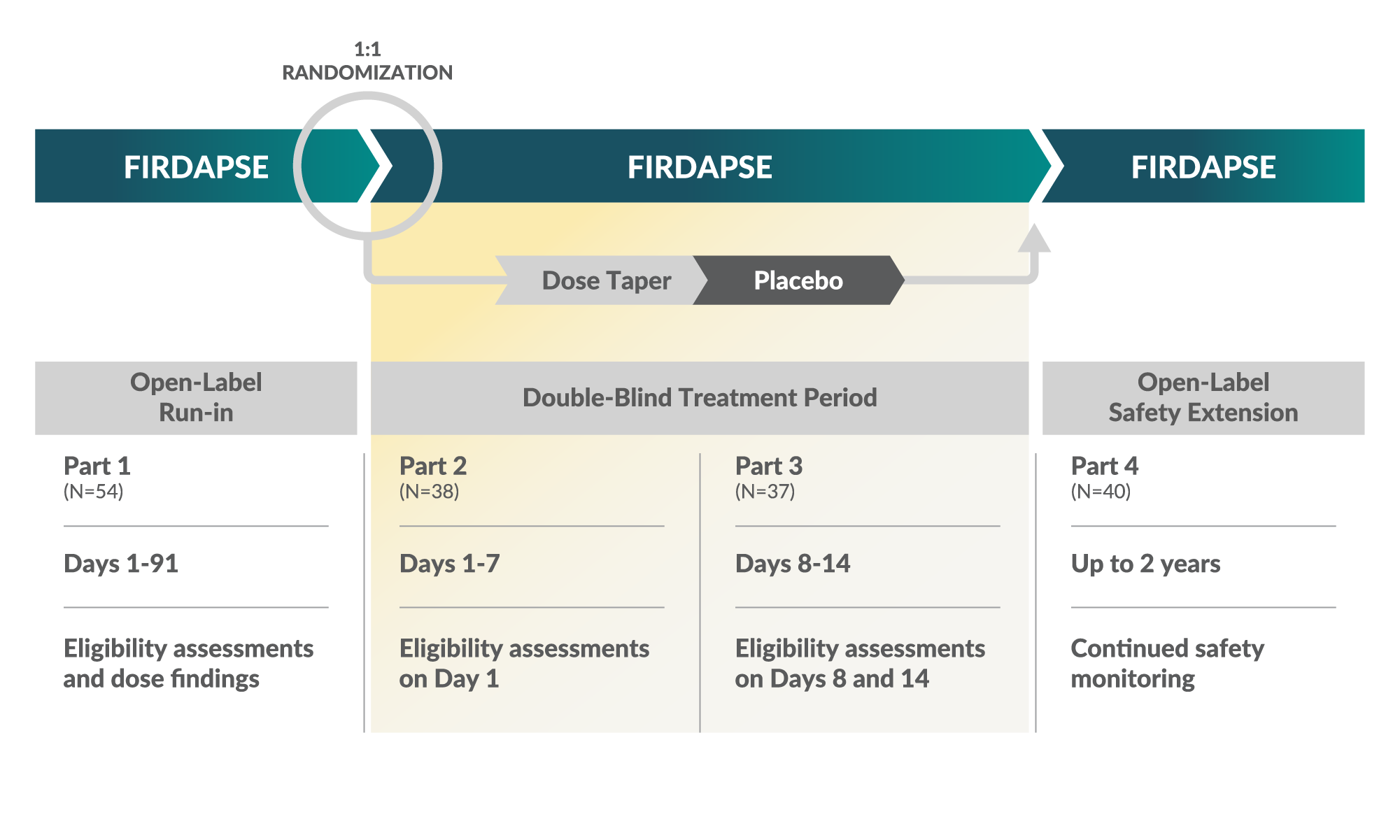
Eligibility1,2
Patients were aged ≥18 years, ambulatory, had a confirmed LEMS diagnosis, with documented acquired proximal muscle weakness and at least 1 of the following:
- Compound muscle action potential that increased >2-fold after maximum voluntary contraction
- Positive anti-P/Q-type VGCC-Ab test
Open-label phase: Patients received an individualized total daily dosage of FIRDAPSE between 15 and 80 mg/day, given in 3 to 4 divided doses with a maximum single dose of 20 mg (total of >91 consecutive days). Patients had to receive between 30 and 80 mg/day to enter the double-blind phase of the study.
TRIAL 2 STUDY DESIGN3
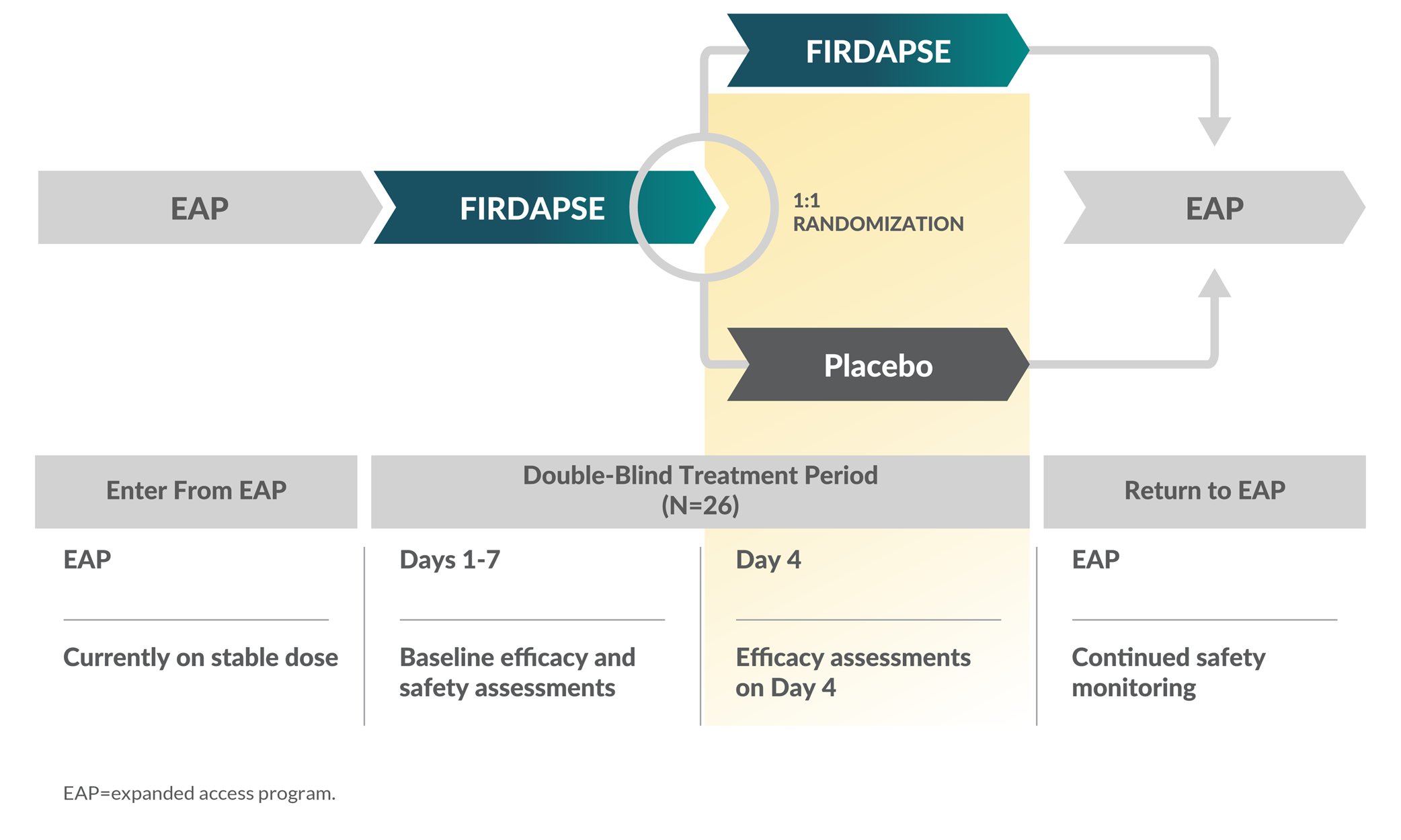
Eligibility1,3
Patients were aged >18 years, ambulatory, had a confirmed LEMS diagnosis, with documented acquired proximal muscle weakness and at least 1 of the following:
- Compound muscle action potential that increased >2-fold after maximum voluntary contraction
- Positive anti-P/Q-type VGCC-Ab test
Dosing: Patients received an individualized total daily dosage of FIRDAPSE between 30 and 80 mg/day, given in 3 to 4 divided doses, with a maximum single dose of 20 mg.

Efficacy and Safety Clinical Trial Data of FIRDAPSE in Patients with LEMS
Take a deeper dive into the study design and clinical findings from the 2 phase 3 studies for FIRDAPSE.