Expand
Indication and Important Safety Information
INDICATIONS AND USAGE:
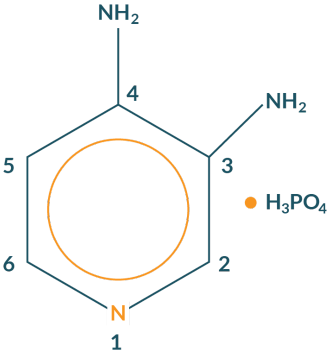
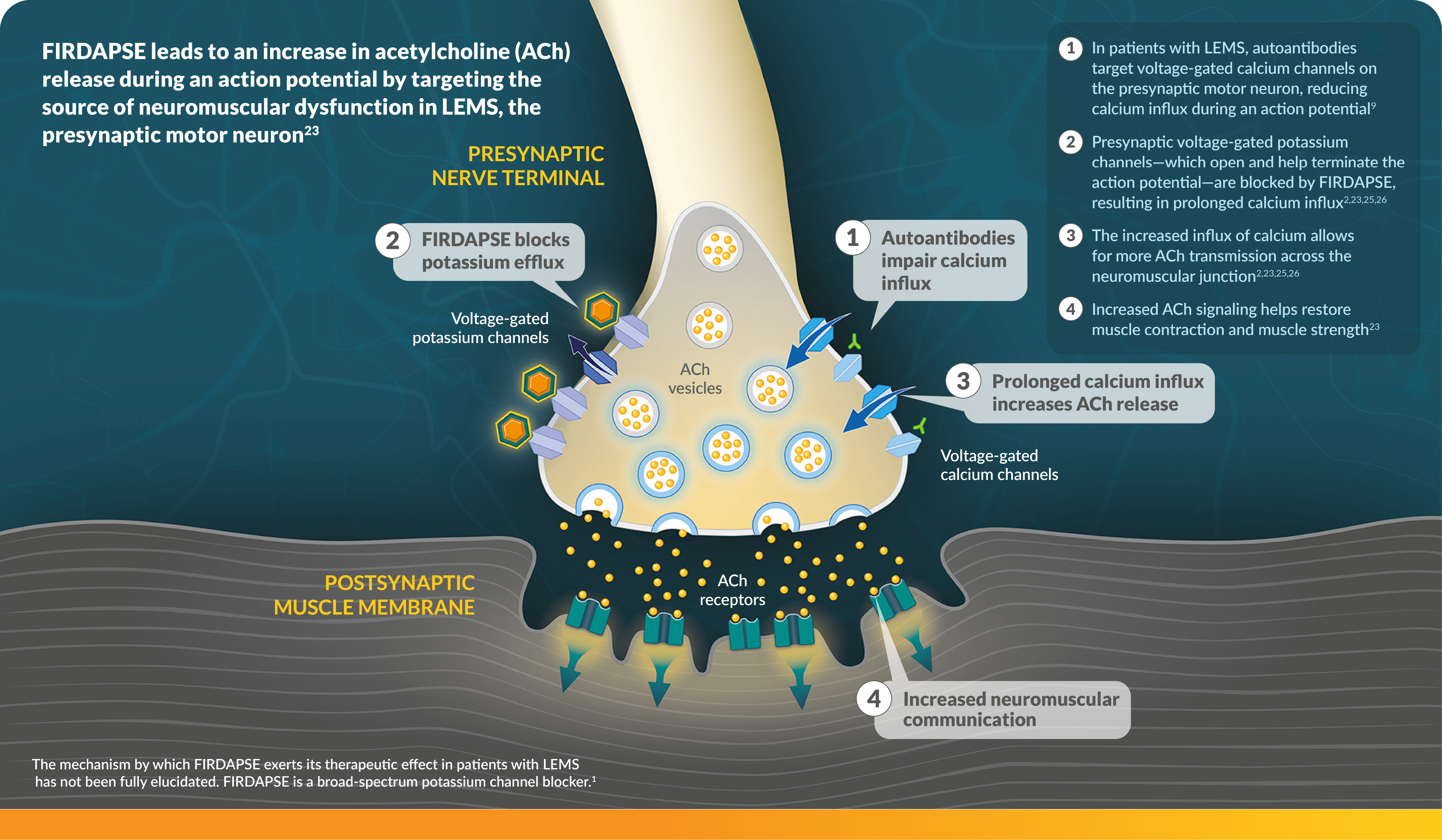
FIRDAPSE is a potassium channel blocker indicated for the treatment of Lambert-Eaton myasthenic syndrome (LEMS) in adults and pediatric patients 6 years of age and older.
CONTRAINDICATIONS
FIRDAPSE is contraindicated in patients with:
- A history of seizures
- Hypersensitivity to amifampridine phosphate or another aminopyridine
WARNINGS AND PRECAUTIONS
Seizures: FIRDAPSE can cause seizures. Consider discontinuation or dose-reduction of FIRDAPSE in patients who have a seizure while on treatment.
Hypersensitivity: If a hypersensitivity reaction such as anaphylaxis occurs, FIRDAPSE should be discontinued and appropriate therapy initiated.
ADVERSE REACTIONS
The most common (> 10%) adverse reactions are: paresthesia, upper respiratory tract infection, abdominal pain, nausea, diarrhea, headache, elevated liver enzymes, back pain, hypertension, and muscle spasms.
To report SUSPECTED ADVERSE REACTIONS, contact Catalyst Pharmaceuticals at 1-844-347-3277 (1-844-FIRDAPSE) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
References: 1. Full Prescribing Information for FIRDAPSE (amifampridine). Catalyst Pharma; 2024. 2. Oh SJ, Scherbakova N, Kostera-Pruszczyk A, et al; LEMS Study Group. Amifampridine phosphate (FIRDAPSE®) is effective and safe in a phase 3 clinical trial in LEMS. Muscle Nerve. 2016;53(5):717-725. 3. Shieh P, Sharma K, Kohrman B, Oh SJ. Amifampridine phosphate (FIRDAPSE) is effective in a confirmatory phase 3 clinical trial in LEMS. J Clin Neuromuscul Dis. 2019;20(3):111-119. 4. Titulaer MJ, Lang B, Verschuuren JJ. Lambert-Eaton myasthenic syndrome: from clinical characteristics to therapeutic strategies. Lancet Neurol. 2011;10(12):1098-1107. 5. Gilhus NE. Lambert-Eaton myasthenic syndrome; pathogenesis, diagnosis, and therapy. Autoimmune Dis. 2011;2011:973808. 6. Kesner VG, Shin JO, Dimachkie MM, Barohn RJ. Lambert-Eaton myasthenic syndrome. Neurol Clin. 2018;36(2):379-394. 7. US Food and Drug Administration. Approved Drug Products With Therapeutic Equivalence Evaluations (Orange Book). 44th ed. 2024:47, Amifampridine; p1944, U-2956. 8. Gandhi L, Johnson BE. Paraneoplastic syndromes associated with small cell lung cancer. J Natl Compr Canc Netw. 2006;4(6):631-638. 9. Tarr TB, Wipf P, Meriney SD. Synaptic pathophysiology and treatment of Lambert-Eaton myasthenic syndrome. Mol Neurolbiol. 2015;52(1):456-463. 10. Drapkin BJ, Morrell DJ, Grebla R, Shechter G, Gerber DE. Marked under-diagnosis of Lambert-Eaton myasthenic syndrome in small cell lung cancer: an analysis of real-world claims data. Front Oncol. 2025;15:1650373. doi:10.3389/fonc.2025.1650373. 11. Bebb DG, Murray C, Giannopoulou A, Felip E. Symptoms and experiences with small cell lung cancer: a mixed methods study of patients and caregivers. Pulm Ther. 2023;9(3):435-450. 12. Harms L, Sieb JP, Williams AE, et al. Long-term disease history, clinical symptoms, health status, and healthcare utilization in patients suffering from Lambert Eaton myasthenic syndrome: results of a patient interview survey in Germany. J Med Econ. 2012;15(3):521-530. 13. Sanders DB. Lambert-Eaton myasthenic syndrome: diagnosis and treatment. Ann N Y Acad Sci. 2003;998:500-508. 14. Kunii E, Owaki S, Yamada K, et al. Lambert-Eaton myasthenic syndrome caused by atezolizumab in a patient with small-cell lung cancer. Intern Med. 2022;61(11):1739-1742. 15. Lipka AF, Boldingh MI, van Zwet EW, et al. Long-term follow-up, quality of life, and survival of patients with Lambert-Eaton myasthenic syndrome. Neurology. 2020;94(5):e511-e520. 16. Briggs SEW, Gozzard P, Talbot DC. The association between Lambert-Eaton myasthenic syndrome and small cell lung carcinoma. Immunotargets Ther. 2013;2:31-37. 17. Titulaer MJ, Lang B, Verschuuren JJ. Lambert-Eaton myasthenic syndrome: from clinical characteristics to therapeutic strategies. Lancet Neurol. 2011; Appendix:1-4. 18. Wirtz PW, Smallegange TM, Wintzen AR, Verschuuren JJ. Differences in clinical features between the Lambert-Eaton myasthenic syndrome with and without cancer: an analysis of 227 published cases. Clin Neurol Neurosurg. 2002;104(4):359-363. 19. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Small Cell Lung Cancer V.2.2026. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed September 26, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way. 20. Motomura M, Lang B, Johnston I, Palace J, Vincent A, Newsom-Davis J. Incidence of serum anti-P/Q-type and anti-N-type calcium channel autoantibodies in the Lambert-Eaton myasthenic syndrome. J Neurol Sci. 1997;147(1):35-42. 21. Schampel A, Kuerten S. Danger: high voltage—the role of voltage-gated calcium channels in central nervous system pathology. Cells. 2017;6(4):43. 22. Strupp M, Teufel J, Zwergal A, et al. Aminopyridines for the treatment of neurologic disorders. Neurol Clin Pract. 2017;7(1):65-76. 23. Lindquist S, Stangel M. Update on treatment options for Lambert-Eaton myasthenic syndrome: focus on use of amifampridine. Neuropsychiatr Dis Treat. 2011;7:341-349. 24. Data on file, Catalyst Pharmaceuticals. 25. Verschuuren J, Strijbos E, Vincent A. Neuromuscular junction disorders. Handb Clin Neurol. 2016;133:447-466. 26. Dodson PD, Forsythe ID. Presynaptic K+ channels: electrifying regulators of synaptic terminal excitability. TINS. 2004;27(4):210-217. 27. Sanders DB, Guptill JT, Aleš KL, et al. Reliability of the triple timed-up-and-go (3TUG) test. Muscle Nerve. 2018;57(1):136-139. 28. Raja SM, Sanders DB, Juel VC, et al. Validation of the triple timed up-and-go test in Lambert-Eaton myasthenia. Muscle Nerve. 2019;60(3):292-298.